Specially designed nanoparticles can reveal tiny cancerous tumors that are invisible by ordinary means of detection, according to a study by researchers at Washington University School of Medicine in St. Louis.
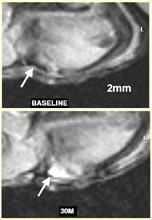
The researchers demonstrated that very small human melanoma tumors growing in mice—indiscernible from the surrounding tissue by direct MRI scan—could be “lit up” and easily located as soon as 30 minutes after the mice were injected with the nanoparticles.
Because nanoparticles can be engineered to carry a variety of substances, they also may be able to deliver cancer-fighting drugs to malignant tumors as effectively as they carry the imaging materials that spotlight cancerous growth.
“One of the best advantages of the particles is that we designed them to detect tumors using the same MRI equipment that is in standard use for heart or brain scans,” says senior author Gregory Lanza, M.D., Ph.D., associate professor of medicine. “We believe the technology is very close to being useful in a hospital setting.”
Lanza and his colleague Samuel Wickline, M.D., professor of medicine, are co-inventors of this nanoparticle technology. The effectiveness of the nanoparticles in diagnosis and therapy in humans will be tested in clinical trials in about one and a half to two years.
The spherical nanoparticles are a few thousand times smaller than the dot above this “i,” yet each can carry about 100,000 molecules of the metal used to provide contrast in MRI images. This creates a high density of contrast agent, and when the particles bind to a specific area, that site glows brightly in MRI scans.
In this study, MRI scans picked up tumors that were only a couple of millimeters (about one twenty-fifth of an inch) wide.
Small, rapidly growing tumors cause growth of new blood vessels, which feed the tumors. To get the particles to bind to tumors, the researchers equipped them with tiny “hooks” that link only to complementary “loops” found on cells in newly forming blood vessels. When the nanoparticles hooked the “loops” on the new vessels’ cells, they revealed the location of the tumors.
Nanoparticles are particularly useful because of their adaptability, according to Lanza, who sees patients at Barnes Jewish Hospital. “We can also make these particles so that they can be seen with nuclear imaging, CT scanning and ultrasound imaging,” Lanza says.
In addition, the particles can be loaded with a wide variety of drugs that will then be directed to growing tumors. “When drug-bearing nanoparticles also contain an imaging agent, you can get a visible signal that allows you to measure how much medication got to the tumor,” Lanza says. “You would know the same day you treated the patient and if the drug was at a therapeutic level.”

Using nanoparticles, drug doses could be much smaller than doses typically used in chemotherapy, making the procedure potentially much safer.
“The other side of that is you have the ability to focus more drug at the tumor site, so the dose at the site might be ten to a thousand times higher than if you had administered the drug systemically,” Lanza says.
The nanoparticles also may permit more effective follow up, because a doctor could use them to discern whether a tumor was still growing after radiation or chemotherapy treatments.
Although this study focused on melanoma tumors, the researchers believe the technology should work for most solid tumors, because all tumors must recruit new blood vessels to obtain nutrients as they grow.

Nevertheless, melanoma has unique traits that make it especially interesting as a target for nanoparticle therapy. Melanoma has a horizontal phase, when it spreads across the skin surface, and a vertical phase, when it goes deep into the body and grows quickly.
“Once melanoma has moved into its vertical phase, it is almost untreatable because by the time the tumors are large enough to detect, it’s too late,” Lanza says. “With the nanoparticles, we believe we would be able to see the smallest melanoma tumors when they are just large enough to begin new blood vessel formation. Plus, we should be able to deliver chemotherapeutic drugs right to melanoma cells, because melanoma tumors create blood vessels using their own cells.”
Schmieder AH, Winter PM, Caruthers SD, Harris TD, Williams TA, Allen JS, Lacy EK, Zhang H, Scott MJ, Hu G, Robertson JD, Wickline SA, Lanza GM. Molecular MR imaging of melanoma angiogenesis with anb3-targeted paramagnetic nanoparticles. Magnetic Resonance in Medicine, 2005;53:621-627.
Funding from the National Institutes of Health, the National Cancer Institute and Philips Medical Systems supported this research.
Washington University School of Medicine’s full-time and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked third in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.