To speed healing at sites of injury – such as heart muscle after a heart attack or brain tissue after a stroke – doctors would like to be able to hasten the formation of new blood vessels. One promising approach is to “mobilize” patients’ blood vessel-forming cells, called angiogenic cells, so these cells can reach the injured area.
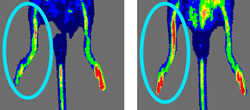
Recently, researchers at Washington University School of Medicine in St. Louis demonstrated that a drug called AMD3100 can mobilize angiogenic cells from bone marrow of human patients in a matter of hours instead of days, as was the case with a related agent called G-CSF.
Angiogenic cells reside mainly in the bone marrow, and when mobilized they can circulate in the bloodstream, homing to sites of injury and helping repair and regrow blood vessels that bring oxygen and nutrients to tissues.
“Like AMD3100, G-CSF can bring these beneficial cells from the bone marrow into the bloodstream, but with G-CSF you don’t see an increase in angiogenic cells until the fourth day,” says senior author Daniel C. Link, M.D., associate professor of medicine in the Division of Oncology. “In a patient who has had a heart attack, that may be too late. In fact, two clinical trials of G-SCF found the treatment doesn’t improve recovery from heart attacks.”
In an article in the journal Blood, the researchers showed that AMD3100 caused a 10- to 20-fold increase in certain angiogenic cells in the blood within four hours in human subjects, suggesting the drug could be a more effective treatment for heart attack or stroke.
The ability of angiogenic cells to enhance recovery from heart attack has become a hot research topic, according to Link, with several clinical trials investigating whether withdrawing bone marrow from a patient and then injecting it at the site of injury will be effective. But Link feels it may make more sense to mobilize the cells into the bloodstream with agents like AMD3100.

“It might be better to let the appropriate cells find their way through the circulation to the site of injury or low oxygen,” he says. “That way you enhance a normal repair mechanism instead of using the brute force of injecting a mixture of many cell types from bone marrow into the affected area.”
The research group has also shown that after treating human subjects with AMD3100 or G-CSF, angiogenic cells can be collected from the blood by a technique that separates them and concentrates them. They found that the cells maintain their function after freezing and so can be stored for future use.
“It’s possible that if a patient was scheduled to have a procedure that would damage blood vessels, such as angioplasty, physicians could collect angiogenic cells before the procedure and use them during the patient’s recovery,” Link says.
Angiogenic cells may also help improve blood flow in peripheral vascular disease, a problem often encountered in diabetic patients, who can suffer from poor circulation in their limbs that causes pain or sores and may lead to amputation.
The potential of the two drugs for treating such conditions was addressed in another study by the research group, also published in a recent issue of Blood, which tested the effect of both AMD3100 and G-CSF in mice after blood flow to a leg had been impaired. Both drugs induced new blood vessels to grow in the affected legs of the mice, and combining the drugs had the greatest benefit.
“We are now working to set up a clinical trial to test G-CSF in patients with peripheral vascular disease,” Link says. “We chose G-CSF in this case because it’s not essential that the treatment act quickly – the condition has been present for months before the patients are treated – and the drug already has FDA approval for medical use. We want to find out if this treatment will lead to long-lasting improvement in blood flow, heal ulcers, relieve pain and prevent amputations.”
Shepherd RM, Capoccia BJ, Devine SM, DiPersio J, Trinkhaus K, Ingram D, Link DC. Angiogenic cells can be rapidly mobilized and efficiently harvested from the blood following treatment with AMD3100. Blood Aug 15, 2006 (electronic publication ahead of print).
Capoccia BJ, Shepherd RM, Link DC. G-CSF and AMD3100 mobilize monocytes into the blood that stimulate angiogenesis in vivo through a paracrine mechanism. Blood May 30, 2006 (electronic publication ahead of print).
Funding from the National Institutes of Health, the Barnes-Jewish Hospital Foundation and Anormed Inc. supported this research.
Washington University School of Medicine’s full-time and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.