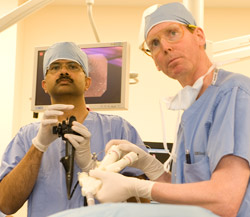
Doctors at Washington University School of Medicine in St. Louis have performed the first non-surgical procedure in the United States that restricts the size of the stomach to treat obesity. The investigational procedure was performed under direct endoscopic visualization with specialized instruments passed into the stomach through the mouth. The first U.S. patient received the treatment on July 23 at Barnes-Jewish Hospital in St. Louis.
 |
|
A new procedure using the TOGA® System allows WUSM physicians to perform a gastroplasty – also known as stomach stapling – through the patient’s mouth. Gastroplasty has proven effective for treating obesity, and this new method requires no surgical incisions.
|
|
|
The procedure was performed as part of the TOGA Pivotal Trial, a multi-center study evaluating an incision-free procedure using the TOGA® System (transoral gastroplasty). Like surgery to treat obesity, the TOGA procedure is designed to alter a patient’s anatomy to give a feeling of fullness after a small meal. The difference is that the investigational technique delivers the treatment through the mouth, without any incisions.
“This is a shift in the way we approach the surgical treatment of obesity,” says Washington University bariatric surgeon J. Christopher Eagon, M.D., who is an assistant professor of surgery and local co-principal investigator for the study. “If this technique provides results close to those achieved with more traditional surgery, it may be an option for people who need to lose a great deal of weight but don’t want to have surgery.”
In the TOGA procedure, the physician introduces a set of flexible stapling devices through the mouth into the stomach, and then uses the staplers to create a restrictive pouch. The pouch catches food as it enters the stomach, giving patients the feeling of fullness after eating less. This study is evaluating the safety and effectiveness of the investigational procedure.
Between February 2006 and July 2007, 47 subjects had the TOGA procedure in a pilot study at medical centers in Mexico and Belgium. They weighed an average of almost 120 pounds over their ideal body weight. Six months after the procedure, subjects had lost more than a third of their excess body weight. By 12 months, their excess weight loss averaged almost 40 percent.
“That’s not as great a loss as we see with gastric bypass surgery, which is still the ‘gold standard’ for weight-loss procedures,” says Washington University gastroenterologist Sreenivasa Jonnalagadda, M.D., who is an associate professor of medicine and co-principal investigator at the St. Louis study site. “The key benefits from an endoscopic procedure as compared to laparoscopic or open surgery are quicker recovery period, shortened hospital stay, decreased risk of complications and an incision-free procedure. And if the restrictive pouch becomes bigger over time, as has been the experience with some obesity surgery patients, it may be possible in the future to further decrease the size of the pouch with this new generation of devices.”
Carrie Williamson of Granite City, Ill., was the first U.S. patient. She was given general anesthesia when receiving the TOGA procedure and stayed overnight at Barnes-Jewish Hospital for observation.
Both Eagon and Jonnalagadda expect the TOGA procedure eventually could be performed on an outpatient basis with sedation rather than general anesthesia, depending on the study’s results.
Subjects will be evaluated regularly for at least one year. All study-related medical care is provided at no charge, and patients receive medically supervised nutrition counseling. Because she was the first, Williamson knew she was getting the actual treatment, but during this study, one of every three volunteers will be a control patient, receiving anesthesia and an endoscopic evaluation of the stomach, without the TOGA procedure, for comparison purposes.
These control patients will be offered the TOGA procedure after 12 months if the procedure proves to be effective. Investigators will evaluate weight loss and monitor obesity-related health problems such as type 2 diabetes, cholesterol levels and hypertension.
“In patients undergoing gastric bypass surgery, it’s common to see blood lipids and blood glucose levels normalize in the days after surgery, even before they lose any weight,” Eagon says. “We may see similar benefits from this procedure, but we need to test that hypothesis.”
The TOGA study will investigate the technique in at least 275 patients at centers across the United States. Investigators at the Washington University Medical Center site are planning to enroll at least 27 volunteers into the trial. Volunteers must be 18 to 60 years old and 100 pounds or more overweight. Some lighter patients may be considered if they have type 2 diabetes or high blood pressure. Patients with a recent heart attack, stroke, chest pain or severe reflux disease are not eligible.
Obesity affects more than 300 million people worldwide according to the World Health Organization. In the United States, the American Society for Metabolic and Bariatric Surgery estimates 205,000 patients had weight-loss surgery in 2007, but that number is estimated to be less than 2 percent of the eligible patient population.
“The TOGA procedure could provide another option for treating obesity,” says Jonnalagadda. “Most of the common treatments — such as diet, exercise and drug therapy — have limited success in this population, and not everyone wants to have surgery, so this may help some patients who haven’t had success with those more traditional treatments.”
For more information about the TOGA study, patients or their families may call Volunteer for Health at 1-866-362-5656 or visit https://vfh.wustl.edu.
Manufactured by Satiety, Inc., the TOGA System is not currently approved by the U.S. Food and Drug Administration.
The TOGA Pivotal Trial is funded by Satiety, Inc.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s Hospitals. The School of Medicine is one of the leading medical research, teaching, and patient care institutions in the nation, currently ranked third in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s Hospitals, the School of Medicine is linked to BJC HealthCare.