
New research suggests that widely used statin therapy provides the most benefit to patients with the highest genetic risk of heart attack. Using a relatively straightforward genetic analysis, the researchers assessed heart attack risk independently of traditional risk factors such as age, sex, so-called good and bad cholesterol levels, smoking history, family history and whether the patient has diabetes.
Patients in intermediate and low-risk categories still benefit from statin therapy, but that benefit is progressively smaller because they’re starting at lower baseline risk, according to the investigators.
The research, from Washington University School of Medicine in St. Louis, Brigham and Women’s Hospital, Massachusetts General Hospital and Harvard Medical School appears March 4 in The Lancet.
For patients at risk of heart disease, doctors routinely prescribe statins, known for their cholesterol-lowering effect. In 2013, the American College of Cardiology and the American Heart Association changed the guidelines for statin therapy, dramatically increasing the number of patients recommended to take it. The move has stirred debate over whether these drugs are overused, especially in light of increasing health-care costs.
“There is ongoing debate over which individuals should be allocated statin therapy to prevent a first heart attack,” said co-first author Nathan O. Stitziel, MD, PhD, a Washington University cardiologist and human geneticist. “Some have said we should be treating more people, while others say we need to treat fewer. As an example of precision medicine, another approach is to identify people at high risk and preferentially prescribe statin therapy to those individuals. Genetics appears to be one way to identify high-risk patients.”
Stitziel noted that this genetic analysis is not available to patients right now. More research is needed to validate the findings before such a test could be developed for clinical use.
Using statistical methods to combine data on 49,000 people enrolled in five studies, the researchers reported that individuals in the high-risk category have a 70 percent higher risk of heart attacks compared with those at lowest genetic risk. They went on to show that statin therapy results in a 13 percent reduction in risk in the low genetic-risk group, a 29 percent reduction in the intermediate group and a 48 percent reduction in the high-risk group.
Stitziel said the new results differ from past research that consistently has shown statins provide about the same relative risk reduction — 30-45 percent depending on dose — across all categories of patients.
“We need more research to confirm these results,” Stitziel said. “Regardless, we can at least say that patients with a high genetic-risk score appear to benefit more from statin therapy because they’re starting at a higher baseline risk, even controlling for all the clinical measures we routinely examine.”
In other words, if a patient has a 10 percent risk of having a heart attack over the next decade and statins cut that risk to 7 percent (a 30 percent reduction), the therapy has a greater absolute benefit than in a patient who starts with a 1 percent risk that is reduced to 0.7 percent (the same 30 percent reduction).
“The panel of genetic markers we analyzed provide a way to identify the patients starting out at higher baseline risk,” Stitziel said. “This is important because it appears to be independent of cholesterol levels and other traditional markers of heart disease that we typically use to estimate risk.”
For each of the risk categories, Stitziel and his colleagues, including co-first author Jessica L. Mega, MD, of Brigham and Women’s Hospital, also calculated the number of patients that doctors would need to treat with statins to prevent one heart attack over a 10-year period. This “number needed to treat” statistic also showed the greater benefit of statins in patients in the high genetic-risk category.
For patients in the lowest risk group, the researchers calculated that doctors would need to treat 57-66 patients for 10 years to prevent one heart attack. In the intermediate risk group, doctors would need to treat 42-47 patients over 10 years to get the same benefit. And in the highest risk group, 20-25 patients would need to take a statin for 10 years to prevent one heart attack.
To calculate the genetic-risk score, the investigators analyzed 27 individual “letters” in each patient’s DNA code. Past work by many different research groups has established significant association between these 27 positions in the genome and risk of coronary heart disease.
Individually, many locations are linked to only a minor increase in risk, and the high-risk letter may be quite common. For example, a T instead of a G in a particular spot may be associated with a 6 percent increase in risk, and that T may be present in 70 percent of the population. But combined, the risk score appears to become a clinically meaningful measure of coronary heart disease risk.
Stitziel also pointed out that these 27 markers do not change with age or lifestyle, so theoretically a person at high risk of developing coronary heart disease could be identified early, before traditional measures of the disease would be detected with a doctor’s physical exam or routine blood work.
Although this approach is currently not available to patients, Stitziel indicated that with additional research this type of genetic-risk score might one day become a useful tool in estimating the degree to which an individual is likely to benefit from statin therapy.
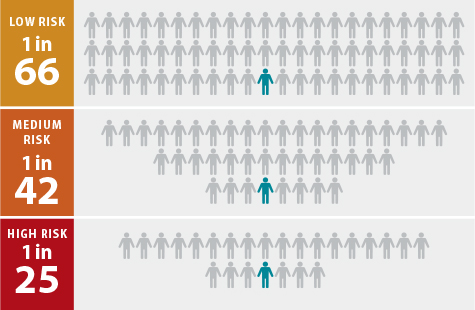
colleagues calculated the number of patients that
doctors would need to treat with statins to prevent one heart attack
over a 10-year period. This “number needed to treat” statistic also
showed the greater benefit of statins in patients in the high
genetic-risk category. As an example, using data from the JUPITER clinical trial (Justification for the Use of Statins in Primary Prevention: an Intervention Trial Evaluating Rosuvastatin), they stratified participants into groups at low, intermediate and high risk of having a heart attack. In the high-risk group, researchers calculated 25 people would need to take a statin for 10 years to prevent one heart attack. Such a strategy could help doctors decide which patients are at high risk and therefore most likely to benefit from statin therapy. (Credit: Eric Young)
Comments and respectful dialogue are encouraged, but content will be moderated. Please, no personal attacks, obscenity or profanity, selling of commercial products, or endorsements of political candidates or positions. We reserve the right to remove any inappropriate comments. We also cannot address individual medical concerns or provide medical advice in this forum.