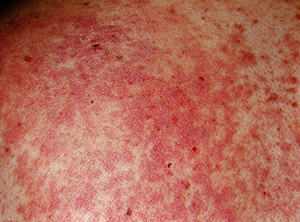
Researchers have designed a way to mitigate graft-versus-host disease, a common and often life-threatening complication of bone marrow transplants that are used to treat leukemia and other blood cancers.
The method, developed at Washington University School of Medicine in St. Louis, also employs a molecular imaging tool to help doctors identify patients most likely to develop this dangerous condition.
In a small pilot study involving eight patients, the technology was shown to be safe. Results are available online in the journal Molecular Therapy.
In leukemia, the body’s white blood cells don’t develop properly, overloading the bone marrow with malignant cells that can then spread throughout the body. Transplants of stem cells from the bone marrow of a donor into these patients is effective at destroying the cancerous cells, but there is a downside.
“Donor immune cells called T-cells are very important, and we’d like them to be able to do their work against the leukemia,” said senior author John F. DiPersio, MD, PhD, the Virginia E. and Sam J. Golman Professor of Medicine. “But they sometimes also attack the skin, gut, liver and other organs, which can create life-threatening complications for patients. We don’t know which patients are likely to develop graft-versus-host disease. We would like to be able to turn off these T cells if they get out of control.”
The pilot study showed that T cells genetically modified to contain a suicide switch can be administered safely to patients. The suicide switch inserted into the donor’s T cells is a gene from the herpes simplex virus. And the switch is triggered by the antiviral drug ganciclovir.
“If donor T cells expand and cause severe graft-versus-host disease, we can give the patient ganciclovir, and it should kill the T cells and stop the process,” said DiPersio, who sees patients at the Siteman Cancer Center at Washington University and Barnes-Jewish Hospital.
According to the study’s first author, Linda G. Eissenberg, PhD, instructor in medicine, the study is unique because it combines the suicide gene technology, which also has been explored by other research groups, with a way to image the donor T cells in the body.
The new imaging component provides a tool to look for clues that might suggest which patients are most likely to develop graft-versus-host disease. According to the investigators, about 40 percent of patients who receive bone marrow transplants experience this potentially dangerous side effect. And the condition is deadly in about a quarter of those who develop it.
“The qualities that make these T cells good at killing leukemia also make them dangerous,” Eissenberg said. “The patients who get graft-versus host disease are more likely to have controlled their leukemia.”
Eissenberg also explained that if they can identify patients most at risk of deadly complications, they can monitor these individuals more closely and provide treatment immediately at the first sign of the dangerous off-target effects. Standard treatments that can control graft-versus-host disease in some patients include steroids.
Past work from DiPersio’s lab has shown that mice with leukemia that are treated with donor T cells and that go on to develop graft-versus-host disease show a characteristic migration pattern of the T cells through the body. When the cells gather early in the thymus, an organ in the neck that plays an important role in immune function, the mice develop graft-versus-host disease. When T cells don’t migrate to the thymus, the mice don’t get the complication.
This early trial was small, with eight patients with relapsed leukemia, each receiving a relatively small number of genetically modified T cells. So the researchers were not able to show whether these migration patterns in mice were similar in people.
But based on the safety results of this study, DiPersio, Eissenberg and their colleagues are participating in a larger trial in partnership with a company based in Italy. The trial will include patients at multiple medical centers, and each participant will receive about 50 times more of the genetically modified donor T cells than were administered in this trial. Washington University is the only center in the trial that will be able to perform the novel imaging studies examining migration patterns of the donated T cells.
“Dr. DiPersio has been interested in reducing graft-versus-host disease for many years,” Eissenberg said. “This is one of his specialties. Right now, giving T cells is the only potentially curative treatment for leukemia, but the therapy itself has a pretty high chance of being deadly. Still, leukemia is so bad you take that risk since, on average, the therapy does extend life.”
Comments and respectful dialogue are encouraged, but content will be moderated. Please, no personal attacks, obscenity or profanity, selling of commercial products, or endorsements of political candidates or positions. We reserve the right to remove any inappropriate comments. We also cannot address individual medical concerns or provide medical advice in this forum.