WashU engineers to help develop rapid COVID-19 test
Researchers at the McKelvey School of Engineering at Washington University in St. Louis received funding for a new COVID-19 test that is based on brand new technology that won’t require brand new tools, making it easy for clinicians to use.
Developing enabling technology for emerging gene therapies
For years, researchers have attempted to harness the full potential of gene therapy, a technique that inserts genes into a patient’s cells to treat cancer and other diseases. However, inserting engineered DNA molecules into cells is difficult. A team of engineers at Washington University in St. Louis has developed a new method that could make the process easier.
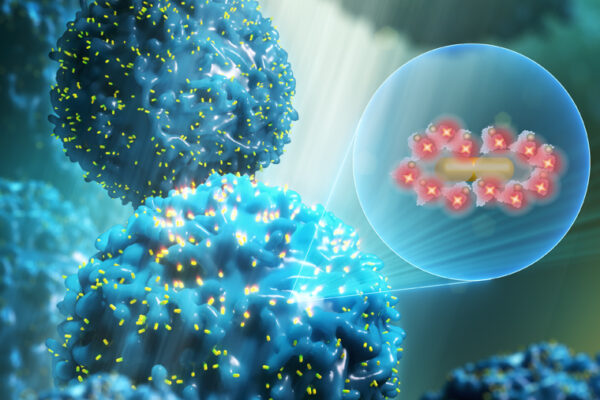
Storing and testing at any temperature
Engineers at Washington University in St. Louis develop new nanoparticle technology that eliminates the need for cold storage in some medical diagnostic tests.
Nanotherapy effective in mice with multiple myeloma
Researchers have designed a nanoparticle-based therapy that is effective in treating mice with multiple myeloma, a cancer of bone marrow immune cells. Targeted specifically to the malignant cells, these nanoparticles protect their therapeutic cargo from degradation in the bloodstream and greatly enhance drug delivery into the cancer cells.
Innovative light therapy reaches deep tumors
Researchers led by Samuel Achilefu, PhD, at the School of Medicine have devised a way to apply light-based therapy to deep tissues never before accessible. Instead of shining an outside light, they delivered light directly to tumor cells, along with a photosensitive source of free radicals that can be activated by the light to destroy cancer.
Nanoparticle that lights up artery-clogging plaque to be evaluated in clinical trial
The Food and Drug Administration (FDA) has approved for testing in people a nanoparticle-based imaging agent jointly developed at the School of Medicine and collaborating institutions. The imaging agent may illuminate dangerous plaque in arteries, and doctors hope to use it to identify patients at high risk of stroke.
Wickline receives chancellor’s innovation award
Samuel A. Wickline, MD, has been chosen to receive the Chancellor’s Award for Innovation and Entrepreneurship at Washington University in St. Louis. He will receive the honor Saturday, Dec. 6. Faculty achievement awards will be presented to David A. Balota, PhD, and Steven L. Teitelbaum, MD.
Nanoparticles treat muscular dystrophy in mice
Researchers at the School of Medicine have demonstrated a new approach to treating muscular dystrophy. Mice with a form of the disease showed improved strength and heart function when treated with nanoparticles loaded with rapamycin, an immunosuppressive drug recently found to improve recycling of cellular waste.
Nanoparticles loaded with bee venom kill HIV
Nanoparticles carrying a toxin found in bee venom can destroy human immunodeficiency virus (HIV) while leaving surrounding cells unharmed, researchers at the School of Medicine have shown. The finding is an important step toward developing a vaginal gel that may prevent the spread of HIV. Shown are nanoparticles (purple) carrying melittin (green) that fuse with HIV (small circles with spiked outer ring), destroying the virus’s protective envelope.
Inverting a standard experiment sometimes produces different results
The standard experimental setup for measuring the cellular uptake of nanoparticles is to place cells in a well on a culture plate and cover them with culture medium containing nanoparticles. The assumption underlying these experiments is that the particles remain well-dispersed. But when a Washington University scientist turned cell cultures upside down, he discovered that this assumption doesn’t always hold. Some experiments preparing for the clinical use of nanoparticles may therefore need to be redone.
View More Stories