Oncologists aren’t sure exactly why patients with the same cancer often respond very differently to the same treatment, but a growing body of evidence suggests the answer lies somewhere in the genes. Now researchers at Washington University School of Medicine in St. Louis have become the first to profile the activity of whole sets of genes involved in processing chemotherapeutic drugs.
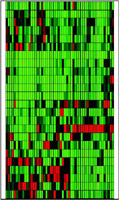
They found that the genes’ activity varied widely among pathologically identical colorectal tumors from different patients. “These findings definitely indicate a need for individualized chemotherapy,” says Howard L. McLeod, Pharm.D., director of the pharmacology core at the Siteman Cancer Center and associate professor of medicine, of genetics, and of molecular biology and pharmacology at the School of Medicine. “Ultimately, our goal is to use this technique to develop a quantitative tool for selecting each patient’s therapy.”
McLeod and colleagues screened tumors from 52 patients with colorectal cancer that had advanced to the stage at which it had spread to nearby lymph nodes. They measured the activity of 24 genes associated with the transport and metabolism of the chemotherapeutic drugs 5-fluorouracil (5-FU) or irinotecan (Camptosar™). Both compounds are commonly used for treatment of colorectal cancer.
The research is reported in two papers in the March 1 and April 1, 2005 issues of Clinical Cancer Research.
Comparing the amount of gene activity among the tumors of each of the 52 patients, the scientists found large differences between individuals. For example, among the genes associated with 5-FU, there was a 16-fold variation in the activity of a gene called FPGS between tumors with the highest and lowest activity levels. In fact, the smallest range of activity among tumors was seen in a gene called DUT, which nevertheless varied by 3-fold.
Using information about variation in gene activity, the researchers identified two distinct groups of patients with similar gene activity profiles in the 5-FU-related gene set. For the Camptosar gene set, they identified three groups of patients.

“We believe the clinical outcomes for each of these patient groups would be very different,” McLeod says. “This type of laboratory analysis can suggest which treatment is appropriate for each patient group in terms of how well they will tolerate the treatment and how the treatment will affect their cancers. Then, clinical studies of the patient groups will allow us to test our hypotheses about the likely response of patients with different gene profiles.”
They also compared the level of gene expression in the tumors and in adjacent non-malignant tissue. The scientists found that 14 out of the 24 genes associated with each drug were either significantly more active or significantly less active in tumors than in normal tissue. The other 10 genes showed little difference in activity.
Gene activity levels in tumors versus normal tissue can provide insight into the fate of chemotherapeutic agents under the influence of the abnormal metabolism of tumors. This information may suggest which genes are likely to make good drug targets and which may be the source of toxic reactions.
Physicians have found that the degree of difference between cancer patients’ reactions to chemotherapy drugs can be enormous. But no method currently exists for an oncologist to accurately predict a patient’s response, and important time is lost when initial treatments fail.
“Say you give Camptosar to a group of 100 people with colorectal cancer,” McLeod says. “You won’t know beforehand which ones, but about 10 of those people will have a severe toxic reaction to the drug, which may even kill them. In about 20 people, the tumors will shrink by half or disappear completely. In another group of 20, Camptosar will have no effect at all on the rate of tumor growth. The rest will have a reaction somewhere in between.”
Several research groups, including McLeod’s, have studied single genetic factors that influence cancer development and treatment. The BRCA markers for increased risk of breast cancer are one such example. But analyzing the activity of a single gene may explain only a small part of the variability in cancers, according to McLeod.
“It’s no surprise that a disease like cancer—where there are multiple genes with abnormal activity—is going to need a solution that addresses multiple genes,” McLeod says. “Our studies will allow us to put relative weights on particular genes. It may be that one gene explains 10 percent of the variation in response to therapy, the next one 15 percent and another one only 2 percent. Knowing the relative contribution of each gene, we can pick the right set of genes to target and have a much better chance of a good outcome.”
To make the connection between their findings on variation in gene activity and the effect of chemotherapeutic drugs on patients, the research team has initiated plans for clinical studies.
“The clinical trials will enable us to identify groups of patients with similar genetic activities related to processing chemotherapeutic drugs,” McLeod says. “After we separate the patients into groups, we can compare each group’s response to the chemotherapy. This should clarify the outcomes associated with particular gene activity profiles and help us learn which treatments work best for which patients.”
Yu J, Shannon WD, Watson MA, McLeod HL. Gene expression profiling of the irinotecan pathway in colorectal cancer. Clinical Cancer Research 2005 Mar 1;11(5):2053-2062.
Kidd EA, Yu J, Li X, Shannon WD, Watson MA, McLeod HL. Variance in the expression of 5-fluorouracil pathway genes in colorectal cancer. Clinical Cancer Research 2005 Apr 1;11(7):2612-2619.
Funding from the National Institutes of Health, the Pharmacogenetics Research Network and the Siteman Cancer Center supported this research.
Washington University School of Medicine’s full-time and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked third in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.