Jill B. Firszt, Ph.D., a cochlear-implant specialist at Washington University School of Medicine in St. Louis, was working on her doctoral dissertation when she met with a 47-year-old patient who been deaf in one ear since childhood. The patient was scheduled to get a cochlear implant in her deaf ear because she recently had a tumor operation that destroyed the hearing in her good ear and left it ineligible for an implant. Firszt knew there wasn’t enough information available about cases like this to predict how well the woman would hear with the implant.
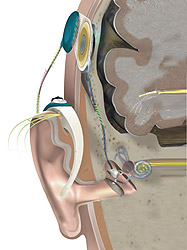
As it turned out, the patient made exceptional progress after her implant surgery. She recognized sentences spoken both in quiet and noise after only three months — much faster than most adults with long-term deafness in both ears who receive cochlear implants.
That patient’s experience inspired Firszt to propose an in-depth study of one-sided or unilateral hearing. She wanted to find out whether hearing in one ear, either natural or because of a cochlear implant, would affect the ability to hear in a deaf ear that receives a cochlear implant. The project has now received a five-year, $3 million grant from the National Institutes of Health (NIH). Collaborators on the project include Rosalie M. Uchanski, Ph.D., research assistant professor of otolaryngology, and Harold Burton, Ph.D., professor of anatomy and neurobiology, of radiology and of cell biology and physiology.
“Patients who have been deaf in both ears for many years often do not achieve the same level of performance as those who have short-term deafness and receive a cochlear implant,” says Firszt, associate professor of otolaryngology and director of the Cochlear Implant Program and Electrophysiology Laboratory. “This patient had long-term deafness in one ear and a short-term deafness in the other. I anticipated that having hearing in one ear for most of her life would improve her outcome with the implant in the long-term deaf ear, but there hadn’t been enough studies to know for sure.”
Normally, each ear sends sounds through a complex auditory system to both the right and left hemispheres of the brain. The sense of sound produced in each hemisphere depends on signals from both ears. That suggests that hearing in only one ear could make it harder for the brain to process some kinds of signals, Firszt says.
On the other hand, even unilateral hearing keeps the right and left hearing centers active to some degree. So having sound through one ear might make it easier for the hearing centers to adapt when they suddenly begin getting signals from the other, formerly deaf, ear through a cochlear implant, Firszt speculates.
Firszt’s NIH-funded project is designed to learn more about the function of the brain’s hearing centers in people with unilateral hearing and subsequent changes when people get cochlear implants.
In one branch of the project, the researchers will conduct studies in bilaterally deaf patients who have one cochlear implant and later get a second implant, and in patients with one deaf ear and one ear with moderate hearing loss who receive an implant in their deaf ear. Patients will be evaluated in real-life listening conditions and followed over time to understand how the hearing in the better ear affects the implanted ear. People with two normal hearing ears will provide a comparison group.
In another branch of the study, people who are deaf in one ear will enable the researchers to obtain more specific information about the kinds of hearing difficulties that unilateral hearing entails. The participants will be evaluated to find out how well they can locate the source of sounds and discern complex sounds like speech mixed with background noise. These same people will undergo brain scans so researchers can study the activity of the hearing centers and correlate brain activity patterns to how people function.
“We’re using imaging techniques such as functional magnetic resonance imaging (fMRI) to look at how different types of sounds are coded in the auditory system,” Firszt says. “When sound input from one ear changes, we expect to see some reorganization of activity in the auditory cortex.”
A second grant, from the Washington University Institute of Clinical and Translational Sciences, will aid in this kind of analysis. This is a one-year, $80,000 grant with potential for continued funding in the second year. It will support research into the time course of brain reorganization when hearing is suddenly lost in one ear. Firszt and colleagues will also implement sound localization training to determine if those with sudden hearing loss can learn to locate sound better, a common problem when a person hears in only one ear.
The results of both studies should provide insight into the consequences of one-sided hearing and help determine how patients can get the maximum hearing improvement from cochlear implants. They should better enable clinicians to counsel patients on what to expect from their implants. The studies should also help determine whether people with bilateral deafness would benefit from implants in both ears and whether it’s best to get a second implant right away or wait a while. People interested in participating in these studies can call (314) 747-0882.
Firszt is also adjunct faculty of the Program in Audiology and Communication Sciences (PACS) at Washington University. The PACS Program is one of a consortium of programs formerly operated by Central Institute for the Deaf, now known as CID at Washington University School of Medicine.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked third in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.
The Washington University Institute of Clinical and Translational Sciences is one of 24 medical research institutions working together as a national consortium to improve the way biomedical research is conducted across the country. The consortium, funded through the Clinical and Translational Science Awards (CTSA), shares a common vision to reduce the time it takes for laboratory discoveries to become treatments for patients, and to engage communities in clinical research efforts. It is also fulfilling the critical need to train the next generation of clinical researchers. The CTSA initiative is led by the National Center for Research Resources at the National Institutes of Health.