In a lecture he delivered in 1906, the German physician Paul Ehrlich coined the term Zauberkugel, or “magic bullet,” as shorthand for a highly targeted medical treatment.
Magic bullets, also called silver bullets, because of the folkloric belief that only silver bullets can kill supernatural creatures, remain the goal of drug development efforts today.
A team of scientists at Washington University in St. Louis is currently working on a magic bullet for cancer, a disease whose treatments are notoriously indiscriminate and nonspecific. But their bullets are gold rather than silver. Literally.
The gold bullets are gold nanocages that, when injected, selectively accumulate in tumors. When the tumors are later bathed in laser light, the surrounding tissue is barely warmed, but the nanocages convert light to heat, killing the malignant cells.
In an article just published in the journal Small, the team describes the successful photothermal treatment of tumors in mice.
The team includes Younan Xia, Ph.D., the James M. McKelvey Professor of Biomedical Engineering in the School of Engineering and Applied Science, Michael J. Welch, Ph.D., professor of radiology and developmental biology in the School of Medicine, Jingyi Chen, Ph.D., research assistant professor of biomedical engineering and Charles Glaus, Ph.D., a postdoctoral research associate in the Department of Radiology.
“We saw significant changes in tumor metabolism and histology,” says Welch, “which is remarkable given that the work was exploratory, the laser ‘dose’ had not been maximized, and the tumors were ‘passively’ rather than ‘actively’ targeted.”
Why the nanocages get hot
The nanocages themselves are harmless. “Gold salts and gold colloids have been used to treat arthritis for more than 100 years,” says Welch. “People know what gold does in the body and it’s inert, so we hope this is going to be a nontoxic approach.”
“The key to photothermal therapy,“ says Xia, “is the cages’ ability to efficiently absorb light and convert it to heat. “
Suspensions of the gold nanocages, which are roughly the same size as a virus particle, are not always yellow, as one would expect, but instead can be any color in the rainbow.

They are colored by something called a surface plasmon resonance. Some of the electrons in the gold are not anchored to individual atoms but instead form a free-floating electron gas, Xia explains. Light falling on these electrons can drive them to oscillate as one. This collective oscillation, the surface plasmon, picks a particular wavelength, or color, out of the incident light, and this determines the color we see.
Medieval artisans made ruby-red stained glass by mixing gold chloride into molten glass, a process that left tiny gold particles suspended in the glass, says Xia.
The resonance — and the color — can be tuned over a wide range of wavelengths by altering the thickness of the cages’ walls. For biomedical applications, Xia’s lab tunes the cages to 800 nanometers, a wavelength that falls in a window of tissue transparency that lies between 750 and 900 nanometers, in the near-infrared part of the spectrum.
Light in this sweet spot can penetrate as deep as several inches in the body (either from the skin or the interior of the gastrointestinal tract or other organ systems).
The conversion of light to heat arises from the same physical effect as the color. The resonance has two parts. At the resonant frequency, light is typically both scattered off the cages and absorbed by them.
By controlling the cages’ size, Xia’s lab tailors them to achieve maximum absorption.
Passive targeting
“If we put bare nanoparticles into your body,” says Xia, “proteins would deposit on the particles, and they would be captured by the immune system and dragged out of the bloodstream into the liver or spleen.”
To prevent this, the lab coated the nanocages with a layer of PEG, a nontoxic chemical most people have encountered in the form of the laxatives GoLyTELY or MiraLAX. PEG resists the adsorption of proteins, in effect disguising the nanoparticles so that the immune system cannot recognize them.
Instead of being swept from the bloodstream, the disguised particles circulate long enough to accumulate in tumors.
A growing tumor must develop its own blood supply to prevent its core from being starved of oxygen and nutrients. But tumor vessels are as aberrant as tumor cells. They have irregular diameters and abnormal branching patterns, but most importantly, they have thin, leaky walls.
The cells that line a tumor’s blood vessel, normally packed so tightly they form a waterproof barrier, are disorganized and irregularly shaped, and there are gaps between them.
The nanocages infiltrate through those gaps efficiently enough that they turn the surface of the normally pinkish tumor black.
A trial run
In Welch’s lab, mice bearing tumors on both flanks were randomly divided into two groups. The mice in one group were injected with the PEG-coated nanocages and those in the other with buffer solution. Several days later the right tumor of each animal was exposed to a diode laser for 10 minutes.
The team employed several different noninvasive imaging techniques to follow the effects of the therapy. (Welch is head of the oncologic imaging research program at the Siteman Cancer Center of Washington University School of Medicine and Barnes-Jewish Hospital and has worked on imaging agents and techniques for many years.)
During irradiation, thermal images of the mice were made with an infrared camera. As is true of other animals that automatically regulate their body temperature, mouse cells function optimally only if the mouse’s body temperature remains between 36.5 and 37.5 degrees Celsius (98 to 101 degrees Fahrenheit). At temperatures above 42 degrees Celsius (107 degrees Fahrenheit) the cells begin to die as the proteins whose proper functioning maintains them begin to unfold.

In the nanocage-injected mice, the skin surface temperature increased rapidly from 32 degrees Celsius to 54 degrees C (129 degrees F).
In the buffer-injected mice, however, the surface temperature remained below 37 degrees Celsius (98.6 degrees Fahrenheit).
To see what effect this heating had on the tumors, the mice were injected with a radioactive tracer incorporated in a molecule similar to glucose, the main energy source in the body. Positron emission and computerized tomography (PET and CT) scans were used to record the concentration of the glucose lookalike in body tissues; the higher the glucose uptake, the greater the metabolic activity.
The tumors of nanocage-injected mice were significantly fainter on the PET scans than those of buffer-injected mice, indicating that many tumor cells were no longer functioning.
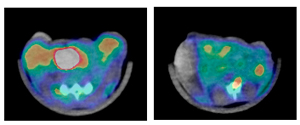
The tumors in the nanocage-treated mice were later found to have marked histological signs of cellular damage.
Active targeting
The scientists have just received a five-year, $2,129,873 grant from the National Cancer Institute to continue their work with photothermal therapy.
Despite their results, Xia is dissatisfied with passive targeting. Although the tumors took up enough gold nanocages to give them a black cast, only 6 percent of the injected particles accumulated at the tumor site.
Xia would like that number to be closer to 40 percent so that fewer particles would have to be injected. He plans to attach tailor-made ligands to the nanocages that recognize and lock onto receptors on the surface of the tumor cells.
In addition to designing nanocages that actively target the tumor cells, the team is considering loading the hollow particles with a cancer-fighting drug, so that the tumor would be attacked on two fronts.
But the important achievement, from the point of view of cancer patients, is that any nanocage treatment would be narrowly targeted and thus avoid the side effects patients dread.
The TV and radio character the Lone Ranger used only silver bullets, allegedly to remind himself that life was precious and not to be lightly thrown away. If he still rode today, he might consider swapping silver for gold.