What have plant scientists learned in the laboratory in the past three to five years that could be used to reduce inputs of water, chemical fertilizers and herbicides to agricultural fields?
This spring, the editors of Science asked Joseph Jez, professor of biology in Arts & Sciences at Washington University in St. Louis, to pull together some of the most interesting recent work in plant science for an article titled, “The Next Green Movement,” published in the Sept. 16 special issue of Science.
The Green Revolution, the wave of innovation in agricultural processes that more than doubled crop yields in the 1960s, relied heavily on dwarf plants that put more of their energy into seed than into stem, chemical fertilizer, agro-chemicals, irrigation and mechanization.
Here, Jez talks about a few of the innovations he chose to highlight in the special issue:
The Green Revolution innovations are basically tapped out, are they not?
In 2013, scientists wrote that global crop production needs to double by 2050 — both because there will be 2 to 3 billion more people to feed, and because diets will shift as more people become middle class. They found that the yields in four key global crops are increasing at rates far too low to meet the projected demand.
Many of the inputs that shored up earlier yield improvements are increasingly untenable because of the energy they use, and the damage they can cause to the environment.
We can’t get by with business as usual. We have to find ways to increase yield while shaving the inputs and lessening the impacts as much as possible.
But we also know far more about plants than we did at the start of the last Green Revolution. Norman Borlaug, the “father” of the Green Revolution, was breeding dwarf varieties of wheat in 1944, nearly a decade before (James ) Watson and (Francis) Crick discovered the structure of DNA.
 When I was a child, my father used a fertilizer on the garden called 10-10-10, which contained 10 percent nitrogen (N), 10 percent phosphate (P2O5) and 10 percent potash (K2O). Nitrogen is often a limiting nutrient, is it not?
When I was a child, my father used a fertilizer on the garden called 10-10-10, which contained 10 percent nitrogen (N), 10 percent phosphate (P2O5) and 10 percent potash (K2O). Nitrogen is often a limiting nutrient, is it not?
There are soil bacteria that can grab it from the atmosphere, but there aren’t enough of them. You can tell it is limiting, because plants grow so much bigger when farmers provide more nitrogen.
We can easily make bio-available nitrogen through chemistry (the Haber process, invented early last century) and that is one of the main reasons we’ve been able to feed 7 billion people.
Estimates are that half of our current food production is traceable to nitrogen fertilizer.
But the Haber process is expensive and energy intensive; it consumes several percent of the world’s annual energy supply, contributing to global warming.
It is also spectacularly wasteful, since 50 to 70 percent of the nitrogen spread on fields is lost to runoff. And nitrogen (and phosphorus) runoff damages the environment, creating, for example, the enormous dead zone in the Gulf of Mexico where the Mississippi River drains into the gulf.
But the plants — some plants at least — have a solution.
Legumes. A quarter of the nitrogen produced is actually produced by legumes, such as soybeans, but also alfalfa, chickpeas, peanuts and clover.

Those plants bring bacteria (rhizobia) into their roots and form little protective nodules, which then become nitrogen-generating factories. It’s a mutually beneficial arrangement; the plants give the bacteria sugar, and the bacteria give the plants nitrogen.
People have been thinking about improving this symbiosis for the past century; which strain of bacteria works best with which plant, gets you the most nodules, the most nitrogen and, in general, the most bang for your buck.
But then folks realized that it might be possible to take the crucial part of the bacteria, the enzyme nitrogenase, which actually makes the nitrogen, and put it into plants, so plants would have the ability to make nitrogen on their own.
We’re getting closer to being able to do that, but there are still a lot of technical hurdles.
Isn’t one problem that nitrogenase is poisoned by oxygen, so you need to put it somewhere special?
Yes, that’s the role of the nodule, to provide a low-oxygen environment for the enzyme. The bacteria are actually infecting the roots of the legume. They’re finding their way in and talking the plant into making them a place to live.
Another option is to decode the signals the plant and bacteria send one another to negotiate the terms of their symbiosis. You could take that process and move it into a non-legumous plant, so that corn would have nitrogen nodules or wheat would have nitrogen nodules. Crop plants would have their own nitrogen-fixing factories.
All plants have signaling systems for recognizing fungi or bacteria. So the signaling system is there. Its just that in the rhizobium/legume interaction, the rhizobium has figured out how to get in and prevent the plant from kicking it out.
We’ve also realized that there are a lot of bacteria that can do nitrogen fixation living in the soil; they just don’t fix nitrogen very efficiently. What if we could modify the nitrogenase in these bacteria to boost their production of nitrogen?
Some plants have yet another method for getting nitrogen, yes?
Yes, they get it from fungi called mycorrhizae that also colonize the roots of plants, pushing fungal threads either between plant cells or into them. The fungal threads transport soil nutrients into the plant and the plant again pays with sugar.

But again, some fungi do this and others don’t, and again, it’s the minority that bring in nitrogen?
Yes, the plants that associate with ectomycorhizzal fungi are not nitrogen limited and can exploit higher carbon dioxide levels in the atmosphere by growing faster. Almost all plants participate in mycorrhizal associations, but only a few percent partner with ectomycorhizae.
But you can start to imagine how you might exploit this ancient mutualism to make nitrogen more available to all plants.
Organic fertilizers help, but they cannot supply the amount of nitrogen needed to feed the world, so biotechnical solutions are looking increasingly attractive.
What about phosphorus? I didn’t know that plants can use only one form of phosphorus.
Yes, orthophosphate (PO4-3), which is why the lawn care company is called Ortho.
I was reading that there isn’t that much of it. You have to get it from mines or guano deposits.
It’s a limited natural resource. A few years ago they were saying the supply would last 20 years, Now its 50 years. The window keeps changing, but the price is going up.
The other part of the problem is that 70 percent of the phosphorus you put on the soil is either consumed by microbes or just trapped in the soil by chemical reactions.
Nitrogen and potassium have been a focus for many years, but people weren’t that interested in phosphorus. But, in the past 10 years, there’s been an explosion in people thinking about this nutrient.
The plants can only use a small part of the phosphorus pool, but there are bacteria that can chewing up a different phosphorus compound, phosphite (PO3-3), that plants can’t metabolize.
What if you took a gene or two from these bacteria, moved it into plants and let plants get orthophosphate from phosphite like the bacteria do?
The cool thing about this is the plant would now be able to use a form of phosphorus that weeds can’t use. So if you fertilized with phosphite, you’d be starving the weeds as well as feeding the plants.
Shall we broach the topic of photosynthesis? I remember when I first learned that plants aren’t really that good at photosynthesis. I was shocked. I wondered how evolution could have created plants that are bad at this. But they get as much energy as they need to survive and reproduce, and there’s no point to gathering more.
Research in photosynthesis is a huge topic, so again, to reduce it to manageable size we tried to look at it from the angle of limiting inputs, such as the amount of land we need for food production.
One idea that we’re working on here is to give plants pigments that can absorb more of the solar spectrum. Plants absorb much of the visible light but little of the infrared and ultraviolet, which means them fail to collect roughly half of the energy the sun throws their way.
But again there are other organisms that we can borrow from?
Yes, algae and some photosynthetic bacteria have photopigments that absorb more of the deep red light, something that would be particularly useful in the lower parts of a plant canopy that receive only light not absorbed by the upper canopy.
Another problem with photosynthesis is that Rubisco, the enzyme that takes the carbon dioxide and fixes it into the molecules that are then combined to make sugar, is incredibly inefficient. Huge amounts of it — 50 percent of the protein in a leaf — are needed to achieve adequate photosynthetic rates.
What’s more it latches onto oxygen as well as carbon dioxide and wastes a lot of energy reacting with oxygen in a process that releases the previously fixed carbon dioxide.
But again you have a small subset of plants that found a way around the oxygen problem.
Yes, the C4 plants, such as corn, sugar cane and millet, physically separate the two processes, concentrating CO2 in microcompartments so that Rubisco can work more efficiently.
There are far fewer C4 than C3 plants, but C4 carbon fixation has independently evolved as many as 60 times in different families of plants.
We know which genes are involved; that’s easy. But we need to figure out is how to get the plant to put the enzyme in a microcompartment; that’s a hard problem.
What about making more efficient Rubisco?
People tried for years to engineer the enzyme, to make it more efficient or reduce its affinity for oxygen. Eventually they turned to screening other organisms for variations of the enzyme that are more efficient.
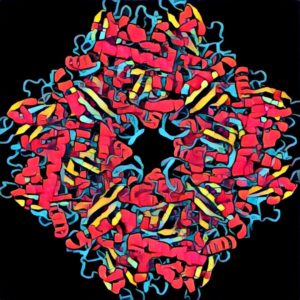
Recently, scientists reported successfully transplanting Rubisco from cyanobacteria into tobacco plants, together with a gene needed to make a microcompartment that keeps oxygen out and carbon dioxide in.
If you can turn the handle faster to get carbon dioxide pulled in faster, you’re also pulling carbon dioxide out of the atmosphere, you’re drawing down this greenhouse gas.
So there is a potential benefit for the environment here, although we don’t yet know how it will work out.
What do you say to people who oppose the genetic engineering of plants?
Think about when people first saw teosinte, the precursor of corn. The people who started harvesting teosinte could never have imagined what corn would become. Corn is a Frankenfood, manipulated to have grotesque traits that are appealing to us.
Now, run the clock forward to 2050. By then, we’ll have another 2 billion people to feed, so another India and another China within the lifespan of our children.
We’re going to have to do what we’ve always done — improve plants for our benefit — just do it in the next 30 years instead of in thousands of years it took to turn teosinte into corn.
The methods by which we modify plants for our purposes are much better controlled now than they were in the past. Traditional selective breeding and interspecies crosses blindly alter thousands to hundreds-of-thousands of genes in a plant. And many foods we eat today were created by exposing seeds to chemicals or radiation to speed up the process. Rust-resistant wheat was made by exposing a chromosome from a rust-resistant grass to X-rays and reincorporating massive chunks of foreign DNA in the wheat.
Modern plant manipulation is mild by comparison. In most GMO foods, only a few well-studied genes are introducing to the plant and the newest techniques, such as CRISPR, allow scientists to precisely edit existing DNA.
