The first two vaccines created with mRNA vaccine technology — the Pfizer/BioNTech and Moderna COVID-19 vaccines — are arguably two of the most effective COVID vaccines developed to date. In clinical trials, both were more than 90% effective at preventing symptomatic infection, easily surpassing the 50% threshold the Food and Drug Administration had set for COVID-19 vaccines to be considered for emergency use authorization.
While breakthrough infections have increased with the emergence of the delta and omicron variants, the vaccines remain quite effective at preventing hospitalizations and deaths. The success of the new technology has led scientists to try to figure out why mRNA vaccines are so effective and whether the protection they provide is likely to endure as new variants arise.
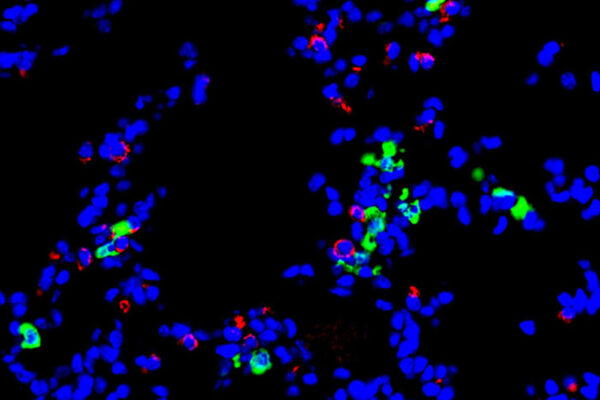
A new study from researchers at Washington University School of Medicine in St. Louis and St. Jude Children’s Research Hospital shines light on the quality of the immune response triggered by mRNA vaccines. The study shows that the Pfizer vaccine strongly and persistently activates a kind of helper immune cell that assists antibody-producing cells in creating large amounts of increasingly powerful antibodies, and also drives the development of some kinds of immune memory. Known as T follicular helper cells, these cells last for up to six months after vaccination, helping the body crank out better and better antibodies. Once the helper cells decline, long-lived antibody-producing cells and memory B cells help to provide protection against severe disease and death, the researchers said.
Further, many of the T follicular helper cells are activated by a part of the virus that doesn’t seem to pick up mutations, even in the highly mutated omicron variant. The findings, published online Dec. 22 in the journal Cell, help explain why the Pfizer vaccine elicits such high levels of neutralizing antibodies and suggests that vaccination may help many people continue producing potent antibodies even as the virus changes.
“The longer the T follicular helper cells provide help, the better the antibodies are and the more likely you are to have a good memory response,” said co-corresponding author Philip Mudd, MD, PhD, assistant professor of emergency medicine at Washington University. “In this study, we found that these T follicular helper cell responses just keep going and going. And what’s more, some of them are responding to one part of the virus’s spike protein that has very little variation in it. With the variants, especially delta and now omicron, we’ve been seeing some breakthrough infections, but the vaccines have held up very nicely in terms of preventing severe disease and death. I think this strong T follicular helper response is part of the reason why the mRNA vaccines continue to be so protective.”
The first antibodies produced in response to an infection or vaccination tend not to be very good. B cells need to go through a kind of boot camp in so-called germinal centers in the lymph nodes before they can produce really powerful antibodies. T follicular helper cells are the drill sergeants of these boot camps. The helper cells provide instruction to the antibody-producing cells on making ever more potent antibodies and encourage those with the best antibodies to multiply and, in some cases, turn into long-lived antibody-producing cells or memory B cells. The longer the germinal centers last, the better and stronger the antibody response.
Earlier this year, Ali Ellebedy, PhD, associate professor of pathology and immunology, of medicine and of molecular microbiology at Washington University, reported that, nearly four months after people had received the first dose of the Pfizer vaccine, they still had germinal centers in their lymph nodes that were churning out immune cells directed against SARS-CoV-2, the virus that causes COVID-19.
In this latest study, Mudd and co-corresponding authors Ellebedy and Paul Thomas, PhD, of St. Jude, aimed to understand the role of T follicular helper cells in producing such a strong germinal center response. The research team also included co-first authors Anastasia Minervina, PhD, and Mikhail Pogorelyy, PhD, postdoctoral researchers who work with Thomas at St. Jude, and others.
The researchers recruited 15 volunteers who each received two doses of the Pfizer vaccine three weeks apart. The volunteers underwent a procedure to extract germinal centers from their lymph nodes 21 days after the first dose, just before the second dose; then at days 28, 35, 60, 110 and 200 after the initial dose. None of the volunteers had been infected with SARS-CoV-2 at the start of the study. The researchers obtained T follicular helper cells from the lymph nodes and analyzed them.
The researchers now are studying what happens after a booster dose and whether changes to T follicular helper cells could explain why people with compromised immune systems, such as those with HIV infection, do not mount a strong antibody response.
Mudd PA, Minervina AA, Pogorelyy MV, Turner JS, Kim W, Kalaidina E, Petersen J, Schmitz AJ, Lei Tingting, Haile A, Kirk AM, Mettelman RC, Crawford JC, Nguyen THO, Rowntree LC, Rosati E, Richards KA, Sant AJ, Klenert MK, Suessen T, Middleton WD, the SJTRC Study Team, Wolf J, Teefey SA, O’Halloran JA, Presti RM, Kedzierska K, Rossjohn J, Thomas PG, Ellebedy AH. SARS-CoV-2 mRNA vaccination elicits a robust and persistent T follicular helper cell response in humans. Cell. December 22, 2021. DOI: https://doi.org/10.1016/j.cell.2021.12.026
This study was funded by Washington University Institute for Clinical and Translational Sciences, grant number UL1TR002345 from the National Center for Advancing Translational Sciences of the National Institutes of Health (NIH); Australian Research Council Laureate Fellowship; Australia’s National Health and Medical Research Council, grant numbers 1173871 and 1194036; American Lebanese Syrian Associated Charities at St. Jude; the Center for Influenza Vaccine Research for High-Risk Populations, contract number 75N93019C00052; the St. Jude Center of Excellence for Influenza Research and Surveillance; contract number HHSN272201400006C; the St. Jude Center of Excellence for Influenza Research and Response, contract numbers 75N93021C00016, U01AI150747, U01AI144616-02S1, and R01AI136514; the National Institute for Allergy and Infectious Diseases (NIAID), grant numbers U01AI141990 and U01AI150747; NIAID Centers of Excellence for Influenza Research and Surveillance, contract numbers HHSN272201400006C and HHSN272201400008C; and NIAID Collaborative Influenza Vaccine Innovation Centers, contract number 75N93019C00051.
Washington University School of Medicine’s 1,700 faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is a leader in medical research, teaching and patient care, and is among the top recipients of research funding from the National Institutes of Health (NIH). Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.