Pappu’s research interests are focused on intrinsically disordered proteins (IDPs), specifically their roles in transcriptional regulation, receptor mediated cell signaling, and cellular stress response. The Pappu lab has pioneered the combined use of polymer physics theories, novel homegrown computational methods, and experiments to probe the functional and phenotypic impacts of IDPs. Professor Pappu’s lab also has a significant effort that is focused on neurodegeneration in Huntington’s and Alzheimer’s diseases. The central goal is to understand how protein aggregation and protein homeostasis pathways collude to give rise to neuronal death as a function of aging.

Rohit Pappu
Gene K. Beare Distinguished Professor
Contact Information
- Phone: 314-935-7958
- Email: pappu@wustl.edu
- Website: Website
Media Contact
In the media
What lava lamps and vinaigrette can teach us about cell biology
Rohit Pappu, the Edwin H. Murty Professor of Engineering
Stories
How do cells take out the trash?
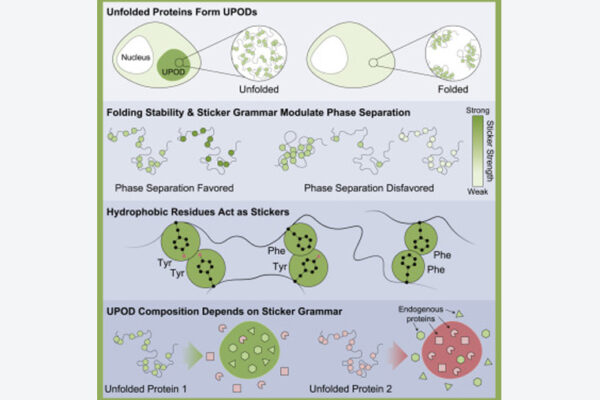
New research led by Kiersten Ruff, a senior research scientist in the lab of Rohit Pappu at the McKelvey School of Engineering, has uncovered the rules that govern how unfolded proteins are identified — and found that exceptions to the rules may play a role in dysfunctional cells.
New structure found in cells
A research group led by Rohit Pappu in the McKelvey School of Engineering and Anthony Hyman at the Max Planck Institute have discovered a new, relevant level of structure in cells.
Pappu lab untangles more IDR secrets
Rohit Pappu’s latest look at intrinsically disordered regions of proteins explains why some sequences behave in different ways. His paper was published in the Proceedings of the National Academy of Sciences.
Understanding features that help cells stay organized
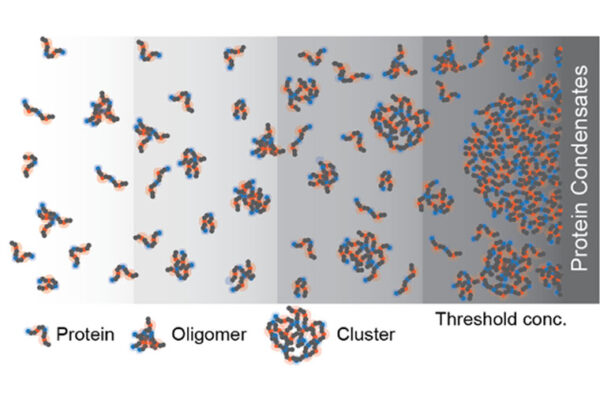
Work from the lab of Rohit Pappu at the McKelvey School of Engineering and colleagues at St. Jude continues to reveal novel findings about phase separation, the process cells use for organization.
International team finds new mechanism critical for formation of membrane vesicles
A collaboration between Washington University in St. Louis, Université de Montréal and McGill University discovers a new importance of biomolecular condensates.
NIH awards $3.1 million grant for Washington University, St. Jude ALS research
Rohit Pappu and collaborator Tanja Mittag received $3.1 to study RNA-binding proteins that are mutated in patients with familial forms of ALS
Pappu, collaborators awarded $7.5 million MURI award
Rohit Pappu, a professor in the McKelvey School of Engineering at Washington University in St. Louis, is part of a multi-institutional team awarded a $7.5 million MURI grant to study and engineer membraneless organelles.
Collaboration lets researchers ‘read’ proteins for new properties
A collaboration between the McKelvey School of Engineering and St. Jude Children’s Research Hospital uncovers the underlying rules that, when broken, contribute to neurodegenerative diseases such as ALS.
Uncovering the design principles of cellular compartments
Membraneless organelles are tiny droplets inside a single cell, thought to regulate everything from division, to movement, to its very destruction. New research from engineers at Washington University in St. Louis uncovers the principles underlying the formation and organization of membraneless organelles.
Detecting diluteness
Engineers at Washington University in St. Louis and Princeton University developed a new way to dive into the cell’s tiniest and most important components. What they found inside membraneless organelles surprised them, and could lead to better understanding of fatal diseases such as cancer, Huntington’s and ALS.
How to stop dividing cancer cells in their tracks
Researchers from Washington University in St. Louis and St. Jude Children’s Research Hospital in Memphis made a discovery that uncovers the molecular logic of how dividing cells are stopped in their tracks. The team zeroed in on a specific protein, whose job is to stop a cell from dividing or to slow the division.
Huntington’s Disease target of $4.5 million in NIH grants
Rohit V. Pappu, the Edwin H. Murty Professor of Engineering in the School of Engineering & Applied Science at Washington University in St. Louis, has received two grants from the National Institutes of Health totaling more than $4.5 million to study the causes behind Huntington’s disease that may ultimately provide clues for a treatment or cure.