As scientists learn more about the key role of inflammation in diabetes, heart disease and other disorders, new research from Washington University School of Medicine in St. Louis suggests that fat in the belly may be an important promoter of that inflammation.
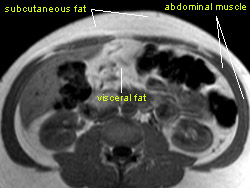
Excess fat is known to be associated with disease, but now the researchers have confirmed that fat cells inside the abdomen are secreting molecules that increase inflammation. It’s the first evidence of a potential mechanistic link between abdominal fat and systemic inflammation.
For years, scientists have been aware of a relationship between disease risk and excess belly fat. “Apple-shaped” people, who carry fat in the abdomen, have a higher risk of heart disease, diabetes and other problems than “pear-shaped” people, who tend to store fat in the hips and thighs. Too much abdominal fat is associated with a defect in the body’s response to insulin. During medical exams, some physicians measure waist circumference to identify patients at increased risk for these problems.
Not just any belly fat will cause inflammation, however. Back in 2004, Washington University investigators found that removing abdominal fat with liposuction did not provide the metabolic benefits normally associated with similar amounts of fat loss induced by dieting or exercising.

“Despite removing large amounts of subcutaneous fat from beneath the skin — about 20 percent of a person’s total body fat mass — there were no beneficial medical effects,” says Samuel Klein, M.D., the Danforth Professor of Medicine and Nutritional Science and the senior investigator on both studies. “These results demonstrated that decreasing fat mass by surgery, which removes billions of fat cells, does not provide the metabolic benefits seen when fat mass is reduced by lowering calorie intake, which shrinks the size of fat cells and decreases the amount of fat inside the abdomen and other tissues.”
In this new study, researchers looked instead at visceral fat — the fat that surrounds the organs in the gut. Unlike subcutaneous fat, visceral fat is not easy to remove surgically because it is very close to the intestines and other internal organs. Since they couldn’t just take out the fat, the research team decided to analyze the blood that ran through it to determine whether visceral fat was involved in inflammation or whether, like subcutaneous fat, it was merely a marker of potential problems.
Reporting in the journal Diabetes, the research team says visceral fat likely contributes to increases in systemic inflammation and insulin resistance. They sampled blood from the portal vein in obese patients undergoing gastric bypass surgery and found that visceral fat in the abdomen was secreting high levels of an important inflammatory molecule called interleukin-6 (IL-6) into portal vein blood.

“The portal vein is filled with blood that drains visceral fat,” says first author Luigi Fontana, M.D., Ph.D., assistant professor of medicine at Washington University in St. Louis and an investigator at the Istituto Superiore di Sanita, Rome, Italy. “Portal vein blood had levels of IL-6 that were 50 percent higher than blood from the periphery.”
Increased IL-6 levels in the portal vein correlated with concentrations of an inflammatory substance called C-reactive protein (CRP) in the body. High CRP levels are related to inflammation, and chronic inflammation is associated with insulin resistance, hypertension, type 2 diabetes and atherosclerosis, among other things.
“These data support the notion that visceral fat produces inflammatory cytokines that contribute to insulin resistance and cardiovascular disease,” says Klein.
Klein, Fontana and J. Christopher Eagon, M.D., assistant professor of surgery, looked at blood samples from 25 patients. All were extremely obese, and all were undergoing gastric bypass surgery. They took blood from the portal vein and from the radial artery in the arm and found differences in levels of IL-6 between the samples.
Fontana believes the findings help explain how visceral fat can lead to inflammation, insulin resistance and other metabolic problems. And he says by contributing to inflammation, visceral fat cells in the abdomen may be doing even more than that.
“Many years ago, atherosclerosis was thought to be related to lipids and to the excessive deposit of cholesterol in the arteries,” Fontana says. “Nowadays, it’s clear that atherosclerosis is an inflammatory disease. There also is evidence that inflammation plays a role in cancer, and there is even evidence that it plays a role in aging. Someday we may learn that visceral fat is involved in those things, too.”
Fontana L, Eagon JC, Trujillo ME, Scherer PE, Klein S. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes, published online Feb. 7, 2007.
This research was supported by grants from the National Institutes of Health.
Washington University School of Medicine’s full-time and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.

