Scientists at Washington University in St. Louis isolated an enzyme that controls the levels of two plant hormones simultaneously, linking the molecular pathways for growth and defense.
Similar to animals, plants have evolved small molecules called hormones to control key events such as growth, reproduction and responses to infections. Scientists have long known that distinct plant hormones can interact in complex ways, but how they do so has remained mysterious.
In a paper published in the Nov. 14 issue of Proceedings of the National Academy of Sciences, the research team of Joseph Jez, professor of biology in Arts & Sciences and a Howard Hughes Medical Institute Professor, reports that the enzyme GH3.5 can control the levels of two plant hormones, auxin and salicylic acid. It is the first enzyme of its kind known to control completely different classes of hormones.
Auxin controls a range of responses in the plant, including cell and tissue growth and normal development. Salicylic acid, on the other hand, helps plants respond to infections, which often take resources away from growth. Plants must tightly control the levels of auxin and salicylic acid to properly grow and react to new threats.
“Plants control hormone levels through a combination of making, breaking, modifying and transporting them,” said Corey Westfall, a former graduate student who led this Jez lab work along with current graduate student Ashley Sherp.
By stitching an amino acid to a hormone, GH3.5 takes the hormones out of circulation, reducing their effect in the plant.
Although scientists suspected GH3.5 controlled auxin and salicylic acid, this double action had not been demonstrated in plants.
“Our question was really simple,” Sherp said. “Can this enzyme actually control multiple hormones? And if that’s true in a test tube, what happens back in a plant?”

To find out, the researchers induced plants to accumulate large amounts of the protein and then measured their levels of hormones. When GH3.5 was expressed at high levels, the amounts of both auxin and salicylic acid were reduced. Deprived of growth-promoting auxin, the plants stayed small and stunted.
The experiment proved that GH3.5 does regulate distinct classes of hormones, but how does it do this?
Searching for answers with X-rays
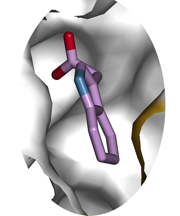
To better understand how the enzyme could control both auxin and salicylic acid, the scientists crystallized GH3.5 and sent the crystals to the European Synchrotron Radiation Facility in Grenoble, France.
The particle accelerator there helped the researchers to fire powerful X-rays into the protein crystal, and the diffraction of the X-rays provided information about the atom-by-atom structure of the enzyme. Westfall assembled this data into a three-dimensional reconstruction of GH3.5, showing it frozen in the act of modifying auxin.
The scientists were expecting to find key differences between GH3.5 and related proteins that would account for its unique ability to modify multiple hormones.
To their surprise, the part of the enzyme that binds and modifies hormones looked almost identical to related enzymes that can only modify auxin. The surprising similarities between the multi-purpose GH3.5 and its single-use relatives suggests that unrecognized elements of these proteins influence which molecules they can bind and transform.
“These surprising results mean there’s something going on that we’re not seeing in the sequence or the structure of these enzymes,” Jez said.
Solving this mystery could tell us more about how enzymes distinguish among similar molecules, a discriminatory ability that is critical for all life, including people as well as plants.

Comments and respectful dialogue are encouraged, but content will be moderated. Please, no personal attacks, obscenity or profanity, selling of commercial products, or endorsements of political candidates or positions. We reserve the right to remove any inappropriate comments. We also cannot address individual medical concerns or provide medical advice in this forum.