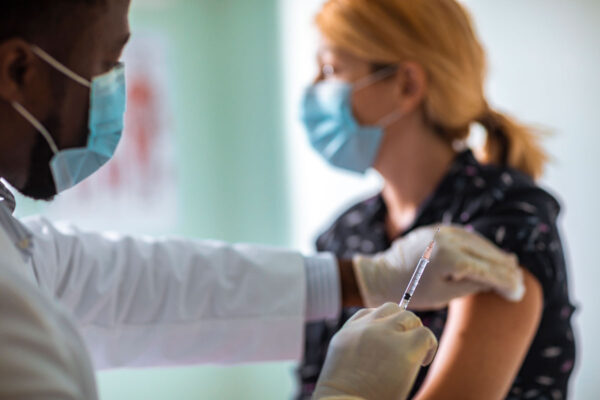
Washington University School of Medicine in St. Louis is a testing site for the Janssen Pharmaceutical Companies of Johnson & Johnson’s phase 3 clinical research study, called the ENSEMBLE trial, to evaluate the safety and efficacy of Janssen’s investigational COVID-19 vaccine candidate, JNJ-78436735, also known as Ad26.COV2.S.
The phase 3 randomized, double-blind, placebo-controlled clinical trial is designed to evaluate the safety and efficacy of a single vaccine dose of the Janssen investigational COVID-19 vaccine candidate versus placebo, in approximately 60,000 adults 18 years old and older, including significant representation from those who are over 60.
“A safe and effective vaccine that can protect against COVID-19 infection is critical for ending the COVID-19 pandemic,” said Rachel Presti, MD, PhD, an associate professor of medicine and medical director of Washington University’s Infectious Diseases Clinical Research Unit, which is leading the trial. “In seeking volunteers, we are hoping to enroll people from diverse communities across the St. Louis region, as well as those who are at high risk of infection due to their age or the nature of their jobs, such as essential workers.”
The School of Medicine will enroll up to 500 adults ages 18 and older in the St. Louis region. The trial will include those both with and without comorbidities associated with an increased risk for progression to severe COVID-19.
To learn more about participating in the COVID-19 vaccine trial at Washington University School of Medicine, please email idcru@wustl.edu or call 314-454-0058 or visit the Division of Infectious Diseases clinical trials website.
ENSEMBLE is being initiated in collaboration with the Assistant Secretary for Preparedness and Response (ASPR), Biomedical Advanced Research and Development Authority (BARDA) and the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH).
Washington University School of Medicine is part of the NIAID-supported COVID-19 Prevention Network (CoVPN). The CoVPN is composed of existing NIAID-supported clinical research networks with infectious disease expertise and is designed for rapid and thorough evaluation of vaccine candidates and monoclonal antibodies for the prevention of COVID-19.
More information about the trial also is available at: www.ensemblestudy.com.
Washington University School of Medicine’s 1,500 faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is a leader in medical research, teaching and patient care, ranking among the top 10 medical schools in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.
Originally published by the School of Medicine
WashU Response to COVID-19
Visit coronavirus.wustl.edu for the latest information about WashU updates and policies. See all stories related to COVID-19.