A new study has prompted scientists to reconsider a once-popular yet controversial idea in stroke research.
Neuroscientists believed that, in the aftermath of a stroke, calming overexcited neurons might prevent them from releasing a toxic molecule that can kill neurons already damaged by lack of oxygen. This idea was supported by studies in cells and animals, but it lost favor in the early 2000s after numerous clinical trials failed to improve outcomes for stroke patients.
But a fresh approach has yielded evidence that the idea may have been discarded too hastily. The new findings are available online in the journal Brain.
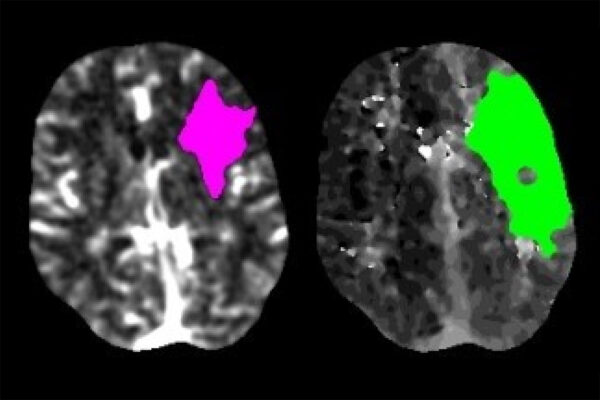
By scanning the whole genomes of nearly 6,000 people who had experienced strokes, researchers at Washington University School of Medicine in St. Louis identified two genes associated with recovery within the pivotal first 24 hours after stroke. Events — good or bad — that occur in the first day set stroke patients on their courses toward long-term recovery. Both genes turned out to be involved in regulating neuronal excitability, providing evidence that overstimulated neurons influence stroke outcomes.
“There’s been this lingering question about whether excitotoxicity really matters for stroke recovery in people,” said co-senior author Jin-Moo Lee, MD, PhD, the Andrew B. and Gretchen P. Jones Professor and head of the Department of Neurology. “We can cure stroke in a mouse using blockers of excitotoxicity. But in humans we performed numerous clinical trials, and we couldn’t move the needle. Every last one of them was negative. In this study, out of 20,000 genes, the top two genetic hits point to mechanisms involving neuronal excitation. That’s pretty remarkable. This is the first genetic evidence that shows excitotoxicity matters in people and not just in mice.”
Every year nearly 800,000 people in the U.S. have ischemic strokes, the most common kind of stroke. Ischemic strokes occur when a clot blocks a blood vessel and cuts off oxygen to part of the brain, triggering sudden numbness, weakness, confusion, difficulty speaking or other symptoms. Over the next 24 hours, some people’s symptoms continue to worsen while others’ stabilize or improve.
In the 1990s, Dennis Choi, MD, PhD, then head of the Department of Neurology at Washington University, performed groundbreaking research on excitotoxicity in stroke. He and others showed that stroke can cause neurons to release large amounts of glutamate, a molecule that transmits excitatory messages between neurons. Glutamate is constantly being released by neurons as part of the normal functioning of the nervous system, but too much all at once can be toxic. Efforts to translate this basic research into therapies for people did not pan out, and eventually pharmaceutical companies let their anti-excitotoxic drug development programs lapse.
But Lee, who formerly worked on excitotoxicity with Choi, did not give up. He teamed up with genetics researcher and co-senior author Carlos Cruchaga, PhD, the Barbara Burton and Reuben M. Morriss III Professor of Neurology and a professor of psychiatry; first author Laura Ibañez, PhD, an assistant professor of psychiatry; and co-author Laura Heitsch, MD, an assistant professor of emergency medicine and of neurology, to tackle the question of what drives post-stroke brain injury. The team identified people who had experienced strokes, and they looked for genetic differences between those who naturally recovered substantial function in the first day and those who did not.
As members of the International Stroke Genetics Consortium, the research team was able to study 5,876 ischemic stroke patients from seven countries: Spain, Finland, Poland, the United States, Costa Rica, Mexico and South Korea. They measured each person’s recovery or deterioration over the first day using the difference between their scores on the National Institutes of Health (NIH) Stroke Scale at six and 24 hours after symptoms first appeared. The scale gauges a person’s degree of neurological impairment based on measures such as the ability to answer basic questions such as “How old are you?”; to perform movements such as holding up the arm or leg; and to feel sensation when touched.
The researchers performed a genomewide association study by scanning the participants’ DNA for genetic variations related to the change in their NIH stroke scale scores. The top two hits were genes that coded for the proteins ADAM23 and GluR1. Both are related to sending excitatory messages between neurons. ADAM23 forms bridges between two neurons so that signaling molecules such as glutamate can be passed from one to the other. GluR1 is a receptor for glutamate.
“We started with no hypotheses about the mechanism of neuronal injury,” Cruchaga said. “We started with the assumption that some genetic variants are associated with stroke recovery, but which ones they are, we did not guess. We tested every single gene and genetic region. So the fact that an unbiased analysis yielded two genes involved in excitotoxicity tells us that it must be important.”
In the years since anti-excitotoxic drug development was abandoned, clot-busting drugs have become the standard of care for ischemic stroke. Such drugs aim to restore blood flow so that oxygen — and anything else in the bloodstream, including medication – can reach affected brain tissue. Consequently, experimental neuroprotective therapies that failed in the past might be more effective now that they have a better chance of reaching the affected area.
“We know that that first 24-hour period has the greatest impact on outcomes,” Lee said. “Beyond 24 hours, there’s diminishing returns in terms of influence on long-term recovery. Right now, we don’t have any neuroprotective agents for that first 24 hours. Many of the original studies with anti-excitotoxic agents were performed at a time when we weren’t sure about the best trial design. We’ve learned a lot about stroke in the last few decades. I think it’s time for a re-examination.”
Ibañez L, Heitsch L, Carrera C, Farias FHG, Del Aguila JL et al. Multi-ancestry GWAS reveals excitotoxicity associated with outcome after ischaemic stroke. Brain. Feb. 25, 2022. DOI: 10.1093/brain/awac080
This work was supported by the Emergency Medicine Foundation; the American Heart Association, grant number 14CRP18860027; the National Institute of Neurological Diseases and Stroke, grant numbers R01-NS085419, R37-NS107230, U24-NS107230 and K23-NS099487; the National Institute on Aging, grant number K99-AG062723; The Foundation for Barnes-Jewish Hospital; Biogen; BrightFocus Foundation; the U.S. Department of Defense; Helsinki University Central Hospital; Finnish Medical Foundation; the government of Finland; Spanish Ministry of Science and Innovation; Instituto de Salud Carlos III, grant number PI051737; Fondos de Investigación Sanitaria ISC III, grant numbers PI10/02064, PI12/01238, PI15/00451 and JR18/00004; Fondos FEDER/EDRF Red de Investigación Cardiovascular, grant number RD12/0042/0020; Fundació la Marató TV3; Genestroke Consortium, grant number 76/C/2011; and Recercaixa’13, grant number JJ086116.
Washington University School of Medicine’s 1,700 faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is a leader in medical research, teaching and patient care, and currently is No. 4 in research funding from the National Institutes of Health (NIH). Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.